二、组织架构(Organizational structure)
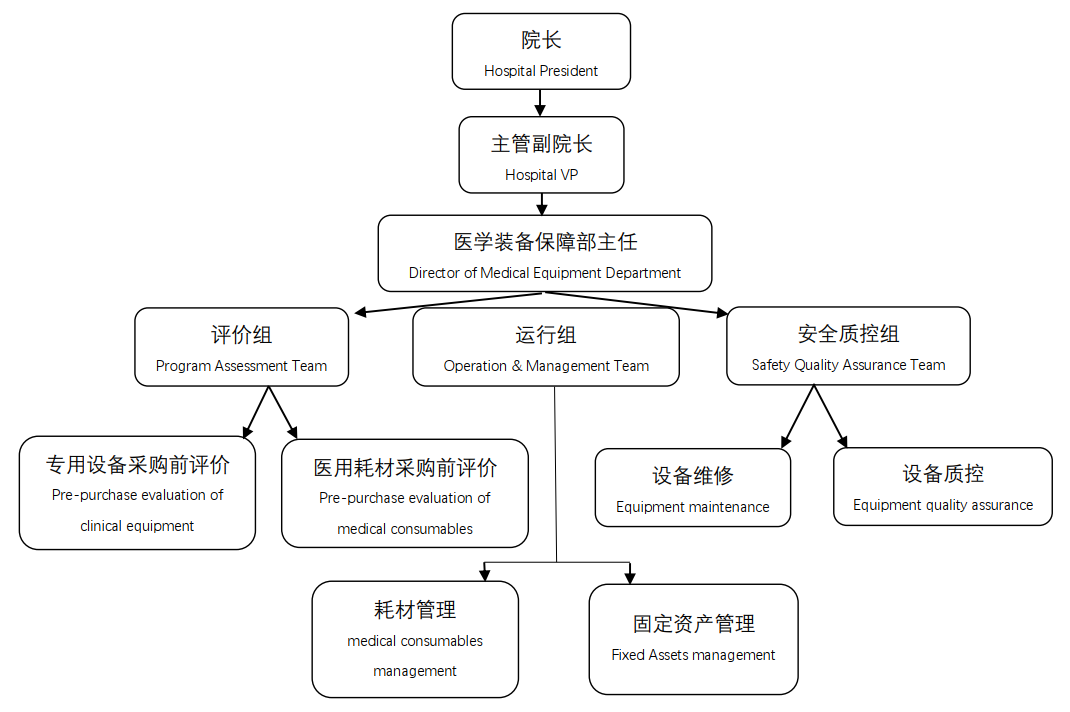
各小组在部门领导的带领下,由组长牵头,实行分级管理,负责全院医疗设备的运行工作,各小组职责内容如下:
安全质控组:
在部门领导的带领下,由组长牵头,负责全院医疗设备的安全运行。对全院医疗设备根据使用风险的高低,制定不同巡检周期;负责全院医疗设备的质控,保障设备有效运行;做好维修记录,整理存档,定期对维修数据做统计、分析,提出新的工作计划和整改意见;定期面向临床对医疗设备进行使用培训。严格按照各自岗位责任制完成工作,坚持原则,按程序办事,尽量避免因设备故障造成医疗事故,保障全院医疗设备的安全运行。
Safety Quality Assurance Team:
During hierarchical management, team leaders take charge of respective work by duties in hospital under the supervision of the department head, the description of each team’s duties has been provided as below:
The quality assurance team makes a well-planned inspection strategy based on the level of risk, assuring the medical instrumentation work properly. To ensure medical equipment work effectively, the quality assurance team has recorded and archived maintenance logs and files, performing periodic statistical analysis of maintenance. Besides, seasonal training for clinical employees has been well-planned to avoid unnecessary human failure in medical equipment operations. The quality assurance team with strict rules-based compliance procedures runs the responsible of respective position-based duties that is so focused on adhering to compliance rules that the team avoid a medical accident caused by medical equipment failures and ensure the safe operation of medical equipment throughout the hospital.
运行组:
在部门领导的带领下,由组长牵头,负责医疗物资进院后的系列管理运行工作,严格按照医院和部门要求,做好物资需求计划、验收、入库、发放、记账、存档及盘点等每一个环节,坚持原则,按程序办事,把好质量关,做好临床物资保障工作。
Operation & Management Team:
The operation and management team is responsible for the series management and operation of medical supplies after they enter into the hospital. According the requirement of hospital and the relevant authorities, diligence is fulfilled in ensuring the requirements planning, acceptance inspection, storage, distribution, accounting, accounting and inventory for medical materials. Upholding the basic work principles, the team act in strict accordance with the relevant rules, fulfill its responsibilities dutifully for the assurance of quality, to better support the clinical practice.
评价组:
在部门领导的带领下,由组长牵头,负责医院年度预算的收集、整理、报送、下发;全院医疗设备、保卫类、信息类、不锈钢产品、试剂耗材等设备物资技术评价(设备物资审批、组织产品介绍、性能对比、功能需求临床和市场调研等);对现有设备物资进行在用情况调研。
Program Assessment Team:
The program assessment team is responsible for the collection, sorting, submission and distribution of the hospital’s annual budget. The technical evaluation covers medical equipment, security program, information software, stainless steel products, reagents and consumables, etc. Also, during standard operating procedure, formal approval of equipment, organization of product introduction, performance comparison, functional requirements, clinical and market research, etc. are a necessary protocol. Additionally, in-use research on current equipment and consumables is also the team’s duty.
